Click here to get an answer to your question ✍️ Calculate the number of protons, electrons, neutrons in Bromine with atomic number 35 and mass number .... Transcribed image text: [Review Topics Use the References to access imp An ion from a given element has 35 protons and 36 electrons. What is the charge on .... Jul 24, 2017 — an anion with 36 electrons, 35 protons and 46 neutrons b. a neutral uranium atom with 143 neutrons in its nucleus c. a cation with a +1 charge ...
- protons electrons and neutrons
- protons electrons and neutrons in oxygen
- protons electrons and neutrons in nitrogen
Feb 26, 2021 — What element has 36 electrons, 38 neutrons, and 35 protons. Protons and neutrons determine the mass of an atom. Continue reading to learn ...
protons electrons and neutrons
protons electrons and neutrons, protons electrons and neutrons in carbon, protons electrons and neutrons in oxygen, protons electrons and neutrons in aluminum, protons electrons and neutrons in nitrogen, protons electrons and neutrons in sodium, protons electrons and neutrons in hydrogen, protons electrons and neutrons calculator, protons electrons and neutrons in chlorine, protons electrons and neutrons in lithium cs7641-machine-learning-midterm-exam-solution
May 3, 2017 — In a neutral atom the number of protons = the number of electrons. 35 protons = 35 electrons. But Bromine anion with a charge of -1 has one extra ... _3d_frog_frenzy_for_mac
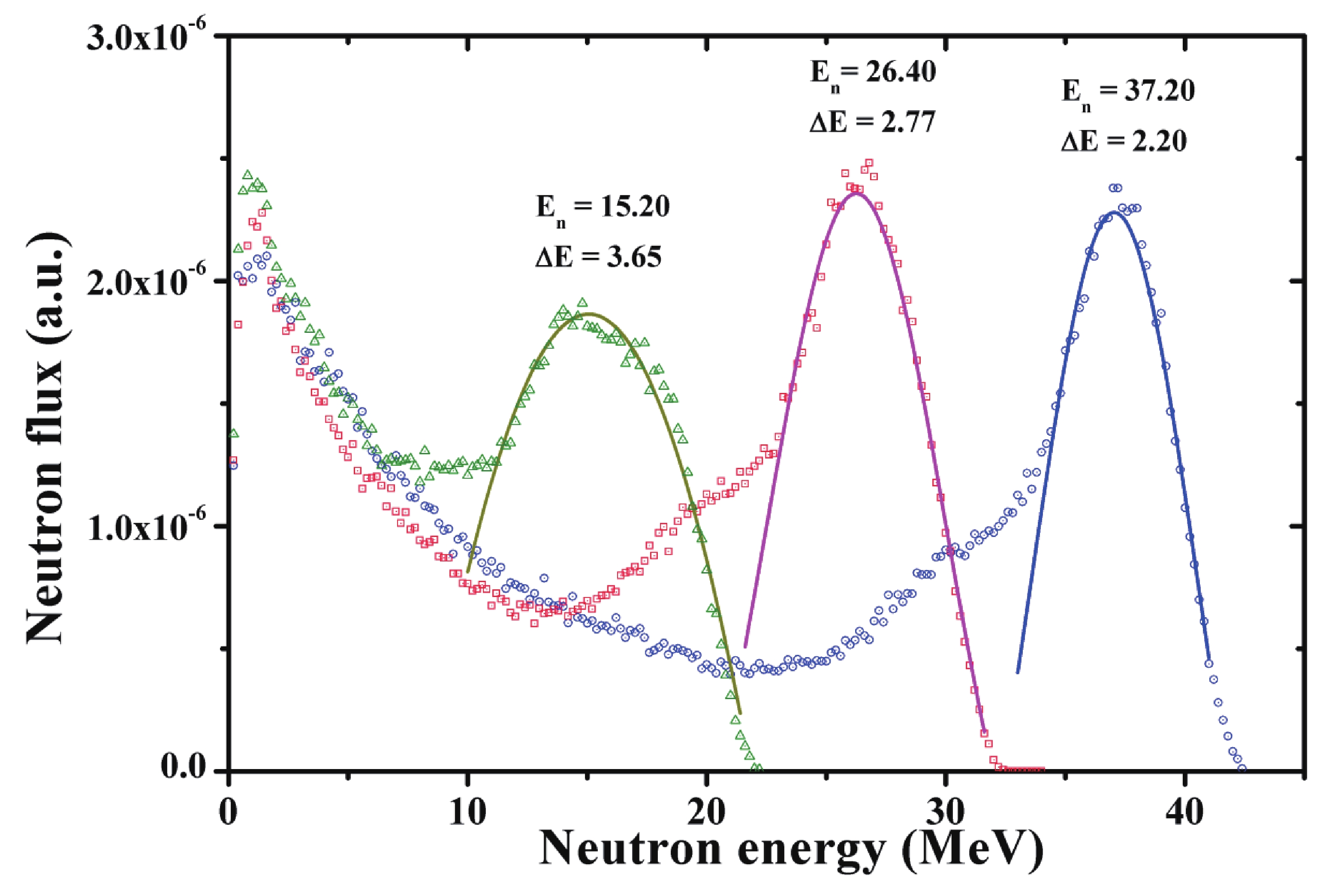
protons electrons and neutrons in oxygen

A bromine ion contains 35 protons, 35 neutrons, and 36 electrons. What is the net charge of the ion? · Question: · Charged Particles: · Answer and Explanation: 1. logilink-ua0072-driver
protons electrons and neutrons in nitrogen
Since there are 35 protons in the nucleus and each proton carries a charge of +1, the charge on the nucleus is +35. The 36 electrons, each with a charge of -1, .... Jun 27, 2010 — An atom with 35 protons will always be bromine. With the information about the neutrons and electrons, you can determine the isotope and the .... Atoms—and the protons, neutrons, and electrons that compose them—are ... 28 electrons, and a mass number of 71; the ion with 36 electrons, 35 protons, and .... What is the symbol of the species with number of electrons equal to 36, protons eual to 35 and neutrons equal to 45 ? More Related Question & Answers.. This tells us that an atom of krypton has 36 protons in its nucleus. ... atom, with 35 protons and 35 electrons, can gain one electron to provide it with 36 electrons.. Write the symbol for each of the following ions: (a) the ion with a 3+ charge, 28 electrons, and a mass number of 71 (b) the ion with 36 electrons, 35 protons, and ... dc39a6609b free printable fairy tale masks
